
Electrolysis in Rechargeable Lead-Acid Batteries
Understanding Electrolysis Effects on Battery Performance and Maintenance
Key Highlights
- Electrolysis Mechanism: During charging, water in the sulfuric acid electrolyte decomposes into hydrogen and oxygen gases.
- Impact on Battery Health: Electrolysis leads to electrolyte water loss and necessitates periodic maintenance.
- Maintenance and Safety: Proper charging protocols and ventilation are vital to prevent overcharging and gas buildup.
Introduction to Electrolysis in Lead-Acid Batteries
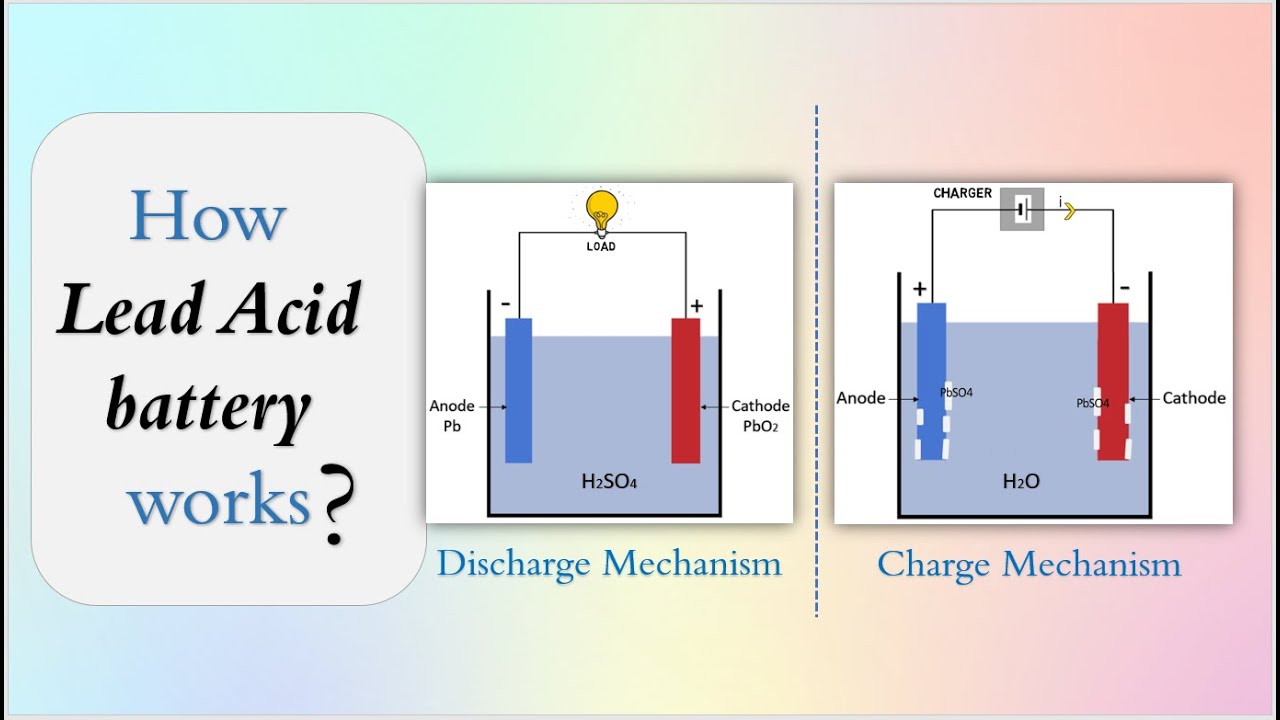
Lead-acid batteries have been in use for over a century and remain widely employed in vehicles, backup power systems, and grid storage applications. One of the fundamental processes occurring in these batteries is electrolysis. Although the notion of electrolysis is traditionally associated with the splitting of water into hydrogen and oxygen in electrolytic cells, a related phenomenon occurs in rechargeable lead-acid batteries, particularly during the charging cycle.
Electrochemical Processes in Lead-Acid Batteries
Discharge and Charge Cycles
In a lead-acid battery, the discharge process involves a chemical reaction between lead dioxide (PbO₂) at the positive electrode, spongy lead (Pb) at the negative electrode, and the sulfuric acid (H₂SO₄) electrolyte. This reaction forms lead sulfate (PbSO₄) and water, releasing electrical energy. During charging, an external voltage is applied, which forces the reverse reactions: lead sulfate is converted back into lead dioxide and spongy lead, restoring the battery’s stored energy.
Reversal of Chemical Reactions
The charging process is analogous to certain electrolytic reactions because it involves driving non-spontaneous reactions using external energy. However, in the context of lead-acid batteries, the primary focus is on restoring the electrode materials rather than performing a traditional electrolysis reaction.
Role and Impact of Electrolysis
Decomposition of Water
While the main charging reactions involve the conversion of lead sulfate back to the active electrode materials, a side reaction occurs in which water molecules present in the electrolyte undergo electrolysis. Under higher charging voltages or overcharging conditions, the water in the electrolyte is split into hydrogen (H₂) and oxygen (O₂) gases.
Electrolyte Water Loss
The generation of hydrogen and oxygen gases results in the loss of water from the electrolyte. This water loss has several implications for battery performance:
- Concentration Increase: The removal of water leads to a higher concentration of sulfuric acid in the electrolyte, potentially altering the battery's chemical balance and affecting both performance and longevity.
- Maintenance Requirement: Flooded lead-acid batteries require regular inspection and refilling with distilled water to maintain optimal electrolyte levels.
- Efficiency Impact: Overcharging or operating at high temperatures accelerates electrolysis, resulting in more rapid water loss, higher gas evolution, and decreased battery efficiency.
Gas Venting and Safety Considerations
The gases produced from the electrolysis, if not properly vented, can accumulate, posing a safety risk due to their flammability, especially in environments with poor ventilation. Lead-acid battery designs often include venting systems to allow these gases to escape safely.
Factors Influencing Electrolysis in Lead-Acid Batteries
Charging Conditions
The rate and extent of electrolysis in lead-acid batteries are influenced by several operational parameters:
- Charging Voltage: Higher voltages accelerate the electrolysis of water, leading to increased gas production and water loss.
- Charging Rate: Fast charging generates more heat and usually results in a higher degree of electrolysis. Maintaining recommended charging rates is essential for battery longevity.
- Ambient Temperature: Elevated temperatures enhance the rate of chemical reactions in the battery, including water electrolysis. Batteries operating in hotter climates may therefore be more prone to electrolyte loss.
- Battery Age: As batteries age, their internal resistance increases, effectively altering the behavior during charging and sometimes exacerbating the effects of electrolysis.
Periodic Maintenance and Monitoring
Given that electrolyte water loss due to electrolysis is an inherent part of the charge cycle, regular monitoring and maintenance are critical:
- Electrolyte Level Checks: Regular inspections help ensure that the electrolyte remains at the correct level. If water loss is detected, distilled water should be added to maintain the proper acid concentration.
- Charging Protocols: Utilizing chargers that are designed to avoid overcharging can reduce excessive electrolysis. These chargers typically employ smart charging cycles that monitor voltage and temperature.
- Ventilation: Ensuring adequate ventilation around battery installations minimizes the accumulation of hydrogen gas and reduces the risk of explosive conditions.
Comparative Analysis and Additional Insights
Electrolysis Versus Regular Charging Functions
It is important to differentiate between the essential charging cycle and the side reaction of electrolysis in a lead-acid battery. While the primary function during charging is the reversal of discharge reactions, the electrolysis of water is a secondary effect that has significant implications for maintenance and safety. This is in contrast to other electrochemical systems, such as water electrolyzers or fuel cells, where the splitting of water is the principal objective.
Implications for Battery Longevity and Performance
The controlled occurrence of electrolysis is fundamental for reliable battery operation. However, when electrolysis is excessive, either due to high charging voltages or improper charging techniques, the unintended effects include:
- Enhanced degradation of the electrolyte composition
- Accelerated water loss demanding more frequent maintenance
- Potential buildup of gas pressure that, if unchecked, could cause physical damage to the battery casing
These factors underscore why proper charging infrastructure and adherence to manufacturer recommendations are pivotal. Monitoring specific parameters during charging can aid in optimizing battery performance and extending operational lifespan.
Summary Table of Key Parameters and Effects
| Parameter | Effect on Electrolysis | Maintenance Implications |
|---|---|---|
| Charging Voltage | Higher voltages increase gas production | Frequent water refills required |
| Charging Rate | Faster charging accelerates water splitting | Increased heat generation; may require cooling measures |
| Ambient Temperature | Elevated temperatures enhance chemical reactions | Risk of accelerated electrolyte loss |
| Battery Age | Aging increases internal resistance | Enhanced sensitivity to overcharging effects |
| Ventilation | Proper venting mitigates gas buildup | Reduces explosion risk and maintains safety |
References
- Electrolysis in Vented Lead Acid Batteries - PWR Storage Solutions
- Loss of Electrolyte - Battery University
- Lead–acid Battery - Wikipedia
- Electrolysis in Commercial Batteries - LibreTexts
- Maintaining Battery Electrolyte Levels - Engineers Edge
Recommended Related Queries
Last updated March 17, 2025