
Complete Overview of RBC Maturation
Exploring every stage from stem cell to mature red blood cell
Key Highlights
- Sequential Development: Erythropoiesis involves a stepwise process with distinct morphological changes.
- Regulatory Mechanisms: Oxygen concentration and hormones like erythropoietin regulate red blood cell production.
- Functional Maturation: The eventual transformation into mature, enucleated, biconcave red blood cells is critical for efficient oxygen delivery.
Introduction to Erythropoiesis
Red blood cell (RBC) maturation, or erythropoiesis, is a complex, highly regulated process occurring predominantly in the bone marrow. It begins with a multipotent hematopoietic stem cell and culminates in the formation of fully mature erythrocytes—cells that are specially designed to transport oxygen throughout the body. This intricate process is orchestrated by a variety of internal signals and external stimuli, ensuring that oxygen delivery is maintained according to the body’s metabolic demands.
The process is initiated when oxygen levels in circulation drop, prompting the kidneys to secrete erythropoietin—a hormone that drives the maturation and amplification of red blood cell precursors. The maturation itself unfolds through a carefully controlled series of stages where cells exhibit distinct morphological and biochemical changes. Over the course of approximately seven days, these developing cells transform in size, nuclear structure, and cytoplasmic composition before emerging as the mature red blood cells that circulate in our bloodstream.
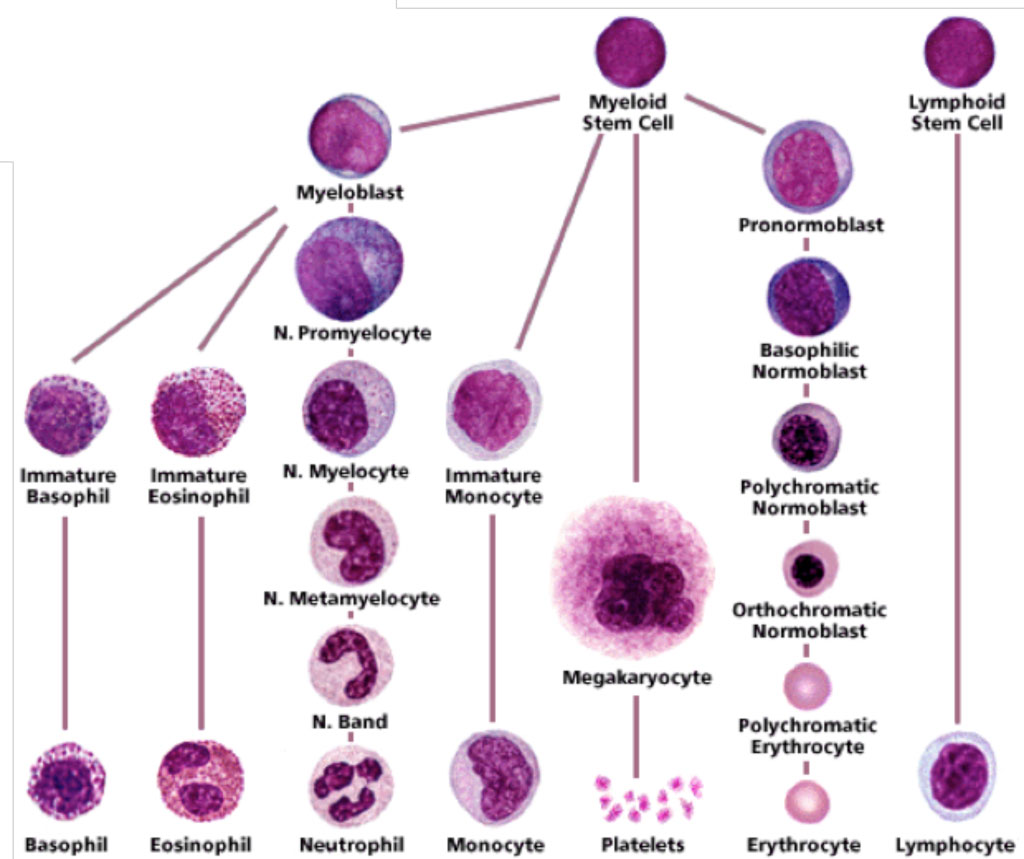
Stages of Red Blood Cell Maturation
From Stem Cell to Proerythroblast
The journey of an RBC begins in the hematopoietic tissue of the bone marrow. It all starts with a hematopoietic stem cell (HSC), a cell with the potential to differentiate into various blood cell types. The differentiation begins when a multipotent HSC commits to becoming part of the erythroid lineage. This occurs after the cell differentiates into a common myeloid progenitor (CMP), which further specifies into a megakaryocyte-erythroid progenitor (MEP). These early transitions set the stage for the specific path known as erythropoiesis.
Proerythroblast Stage
The first stage specific to red blood cells is the proerythroblast stage, sometimes referred to as the rubriblast stage. At this point, the cell is one of the largest in the lineage, with a large nucleus characterized by open chromatin and one or more prominent nucleoli. The cytoplasm of the proerythroblast is basophilic because of an abundance of ribosomal RNA. This stage is pivotal as it represents the commitment of the cell to the erythroid lineage.
Early Erythroblast Stages
Basophilic Erythroblast
Following the proerythroblast stage, the cell enters the basophilic erythroblast phase. During this stage, the cell begins to reduce in size, typically measuring between 10 to 17 micrometers. The nucleus remains round with a high nucleus-to-cytoplasm ratio. The cytoplasm is intensely basophilic—a reflection of the heavy ribosomal content necessitated by active hemoglobin synthesis.
Polychromatophilic (Intermediate) Erythroblast
Next in line is the polychromatophilic erythroblast stage. The cells now display mixed staining characteristics; the cytoplasm shows both a blue (basophilic) tint alongside pink (eosinophilic) regions due to the increasing production of hemoglobin. During this phase, the nucleus begins to condense, preparing for the essential events that occur later in the maturation process.
Orthochromatic Erythroblast (Normoblast)
In the orthochromatic erythroblast stage, also known as the late normoblast stage, the nucleus becomes significantly smaller and more condensed. This stage signals the nearing end of nuclear activity as the cell is primed for enucleation. The cytoplasm is now predominantly eosinophilic, indicating that the hemoglobin synthesis has overtaken other cellular functions. This cytoplasmic staining shift from blue to pink signifies the dominant presence of hemoglobin, which is critical for the cell’s future function in oxygen transport.
Transformation to a Reticulocyte and Maturation
Reticulocyte Formation
One of the most significant steps in erythropoiesis is the expulsion of the nucleus, which transforms the erythroblast into a reticulocyte. This phase is essential because the shedding of the nucleus is the final segmentation that ensures red blood cells are optimized for their primary role in gas exchange. Reticulocytes still contain some residual ribonucleic acid (RNA) and organelles, which can be detected with appropriate staining techniques. Though these cells are considered "immature" in the context of cell development, they are fully capable of synthesizing hemoglobin during this transitional phase.
Reticulocytes undergo further maturation over a short period, typically one to two days, to become fully functional erythrocytes. During this window, any remaining unnecessary organelles are removed, and the plasma membrane is remodeled—often reduced by about 20%. This final cellular reorganization is essential for ensuring that mature red blood cells possess the biconcave shape that supports their flexibility and efficient gas exchange.
Mature Erythrocyte Stage
The end product of erythropoiesis is the mature red blood cell. At this stage, the cell is fully enucleated—lacking a nucleus—and stripped of most organelles such as ribosomes and mitochondria. The mature RBC is characterized by a biconcave disc shape, which maximizes the surface area relative to volume, an ideal characteristic for oxygen transport. With its reduced size and increased hemoglobin concentration, the cell efficiently facilitates the movement of oxygen from the lungs to tissues throughout the body.
Mature red blood cells typically have a lifespan of about 100 to 120 days, after which they are identified for clearance and phagocytosis by macrophages. This removal is crucial as it prevents the accumulation of old or damaged cells in circulation, thereby maintaining overall blood homeostasis.
Detailed Comparison Table of RBC Maturation Stages
| Stage | Key Features | Cell Size (Approximate) | Cytoplasmic Color |
|---|---|---|---|
| Proerythroblast | Large size, open chromatin, prominent nucleoli, high N:C ratio | 17-24 μm | Pale basophilic (blue) |
| Basophilic Erythroblast | Reduced size, intense basophilia, active hemoglobin synthesis | 10-17 μm | Intensely blue |
| Polychromatophilic Erythroblast | Transition stage with mixed staining, increasing hemoglobin | 10-12 μm | Mix of blue and pink |
| Orthochromatic Erythroblast | Compacted nucleus, predominance of hemoglobin, preparation for enucleation | 8-10 μm | Pinkish/red |
| Reticulocyte | Nucleus expelled, residual RNA, transitional phase | Slightly larger than mature RBC | Pink with reticulated appearance |
| Mature Erythrocyte | Enucleated, biconcave, high hemoglobin content, optimized for gas exchange | Approximately 7-8 μm | Salmon/pink |
Regulation of Erythropoiesis
Hormonal Control
A critical factor that regulates red blood cell maturation is the hormone erythropoietin (EPO). Secreted predominantly by the kidney in response to low oxygen levels in the blood, erythropoietin acts upon the bone marrow to stimulate the production and differentiation of red blood cell precursors. This hormonal feedback is crucial for maintaining adequate oxygenation in body tissues, especially under conditions such as high altitude or during chronic hypoxia.
Nutritional Factors
In addition to hormonal regulation, erythropoiesis is highly dependent on a range of nutritional factors. Essential vitamins such as B12 and folate play a vital role in the proper maturation of red blood cells by contributing to DNA synthesis and repair during cell division. Deficiencies in these nutrients can result in ineffective erythropoiesis, often manifesting as various forms of anemia. Adequate levels of iron are also necessary for hemoglobin synthesis, the protein that imparts red blood cells with their oxygen-carrying capacity.
Oxygen Sensing and Feedback Mechanisms
The process of erythropoiesis is tightly linked to the body’s oxygen demands. When oxygen levels drop, the kidney increases the production of erythropoietin, effectively accelerating red blood cell production. Once oxygen levels are restored, erythropoietin levels decrease, leading to a reduction in red blood cell production. This regulatory feedback mechanism ensures that the body does not produce an excess of red blood cells, which could thicken the blood and lead to complications.
Morphological and Biochemical Transitions
Nuclear Changes
A prominent feature of red blood cell maturation is the progressive reduction in nuclear size and complexity, culminating in the expulsion of the nucleus in reticulocytes. Initially, early erythroblasts possess a large nucleus with a high nucleus-to-cytoplasm ratio, reflecting considerable transcriptional activity. As maturation proceeds, chromatin condensation reduces the nuclear volume, ultimately resulting in an enucleated cell. This loss of the nucleus is an essential adaptation, as it not only streamlines the cell for efficient gas exchange but also forces the cell to rely solely on glycolytic metabolism.
Cytoplasmic Transformation
The cytoplasm of developing red blood cells undergoes profound changes throughout the maturation process. In the early stages, the cytoplasm is basophilic due to the high ribosome content required for synthesizing large quantities of hemoglobin. As the cell starts to fill with hemoglobin, the cytoplasm gradually shifts towards an eosinophilic appearance. This transition reflects the strong presence of hemoglobin, which is central to the cell’s eventual role in oxygen transport.
Functional Adaptation
Ultimately, the transformation that takes place during erythropoiesis equips the mature red blood cell with unique functional properties. The biconcave disk shape—resulting from the loss of internal organelles and a reduction in cell volume—imparts the red blood cells with an increased surface area-to-volume ratio. This adaptation is critical for the efficient diffusion of oxygen and carbon dioxide. Moreover, the mature cell’s structural features allow it to deform as it traverses narrow capillaries, ensuring that oxygen delivery is maintained even in the smallest vessels.
Clinical Relevance and Implications
Anemia and Defective Erythropoiesis
Disruptions in the maturation of red blood cells may lead to a variety of hematologic disorders, most notably anemia. This condition is characterized by insufficient levels of functional red blood cells, resulting in decreased oxygen delivery to tissues and consequent fatigue, weakness, or other systemic symptoms. Anemia can arise from nutrient deficiencies, such as iron, folate, or vitamin B12, or due to genetic or acquired defects in the erythropoietic pathway.
In clinical settings, understanding the stages and regulation of erythropoiesis is crucial. It not only aids in diagnosing various types of anemia but also helps in determining appropriate treatment strategies. For instance, patients with anemia due to chronic kidney disease might be treated with synthetic erythropoietin to stimulate red blood cell production, thereby alleviating symptoms related to low oxygen levels.
Monitoring and Research
Modern research often focuses on the detailed cellular and molecular mechanisms underpinning erythropoiesis. With advances in imaging techniques, molecular biology, and bioinformatics, researchers continue to uncover the intricacies of red blood cell development. These insights not only contribute to our understanding of basic biology but also provide avenues for therapeutic interventions in various hematologic conditions.
Integrative Summary of the RBC Maturation Process
The process of erythropoiesis is a highly coordinated sequence of developmental stages, beginning with a multipotent hematopoietic stem cell and culminating in a mature, fully functional red blood cell. Each stage—from the proerythroblast to the reticulocyte—provides essential changes that prepare the cell for its ultimate role in oxygen transport.
Morphological changes, such as reduced cell size, nuclear condensation, and the eventual expulsion of the nucleus, work in tandem with biochemical adaptations like hemoglobin synthesis. The crossover from a basophilic to a more eosinophilic cytoplasm underscores the functional transformation that the cell undergoes. Supplementing these cellular modifications, regulatory mechanisms—both hormonal and nutritional—ensure that erythropoiesis proceeds efficiently, responding to the body’s dynamic oxygen needs.
As newly formed reticulocytes enter the bloodstream, they complete their maturation process by extruding any remaining organelles and acquiring their characteristic biconcave shape. This shape is not merely a structural curiosity; it is critical for optimal gas exchange and the mechanical flexibility required for passage through the narrowest capillaries.
Overall, the maturation of red blood cells is an exemplary model of how cellular development, precise regulation, and functional adaptation converge to meet the physiological demands of an organism. This well-orchestrated progression ensures that our tissues receive a continuous, regulated supply of oxygen, which is vital for all bodily functions.
Conclusion and Final Thoughts
In conclusion, the maturation of red blood cells—erythropoiesis—is a meticulously regulated process essential for maintaining homeostasis and supporting the metabolic needs of the body. Beginning with a multipotent hematopoietic stem cell, each stage of development—from the proerythroblast, through the various erythroblast phases, to the reticulocyte and finally the mature erythrocyte—is characterized by specific morphological and biochemical changes. These changes are imperative for the cell’s transition to a state where it can efficiently transport oxygen.
Critical regulatory factors, such as erythropoietin and essential nutrients like vitamin B12, folate, and iron, ensure that this process is both responsive and adaptive to the body’s oxygen demands. Furthermore, the morphological transformations—especially the enucleation and the formation of a biconcave disc—are vital for achieving the functional properties required for efficient gas exchange and circulation through the body’s complex vascular network.
Understanding the complete process of RBC maturation not only provides insight into normal physiology but also has significant clinical implications. Disruptions in this tightly controlled process can lead to various anemias and other hematologic disorders, underscoring the importance of balanced erythropoiesis in overall health. The study of this dynamic process continues to inform both basic biological research and clinical practice, inspiring novel therapeutic strategies and refined diagnostic tools.
References
- Stages of RBC Maturation Flashcards - Quizlet
-
Erythropoiesis: What It Is & Process Stages - Cleveland Clinic
- Erythropoiesis - Wikipedia
- Red Blood Cell Formation | Process, Stages & Life Cycle - Study.com
-
Erythropoiesis Overview - ScienceDirect
Recommended Further Exploration
Last updated February 27, 2025