
Temporal Interference Stimulation for Parkinson's Disease
Exploring a non-invasive approach to alleviate motor symptoms in PD
Key Highlights
- Non-Invasive Deep Brain Stimulation: TIS enables targeted modulation of deep brain structures without surgery.
- Symptom Alleviation: Early clinical evidence indicates improvements in tremor, bradykinesia, and overall motor control.
- Personalized Modulation: Adaptable stimulation protocols allow treatment customization based on patient-specific needs.
Introduction
Parkinson's Disease (PD) is a progressive neurodegenerative disorder distinguished by motor impairments such as tremors, bradykinesia (slowness of movement), rigidity, and postural instability. Traditional treatment methodologies include pharmacological interventions and deep brain stimulation (DBS). While DBS has shown significant benefits, its invasive nature restricts its use primarily to advanced PD cases. In recent years, a novel approach known as temporal interference stimulation (TIS) has emerged as a potential non-invasive alternative, presenting promising early results in alleviating motor symptoms for individuals with early-stage PD.
Understanding Temporal Interference Stimulation (TIS)
Mechanism of Action
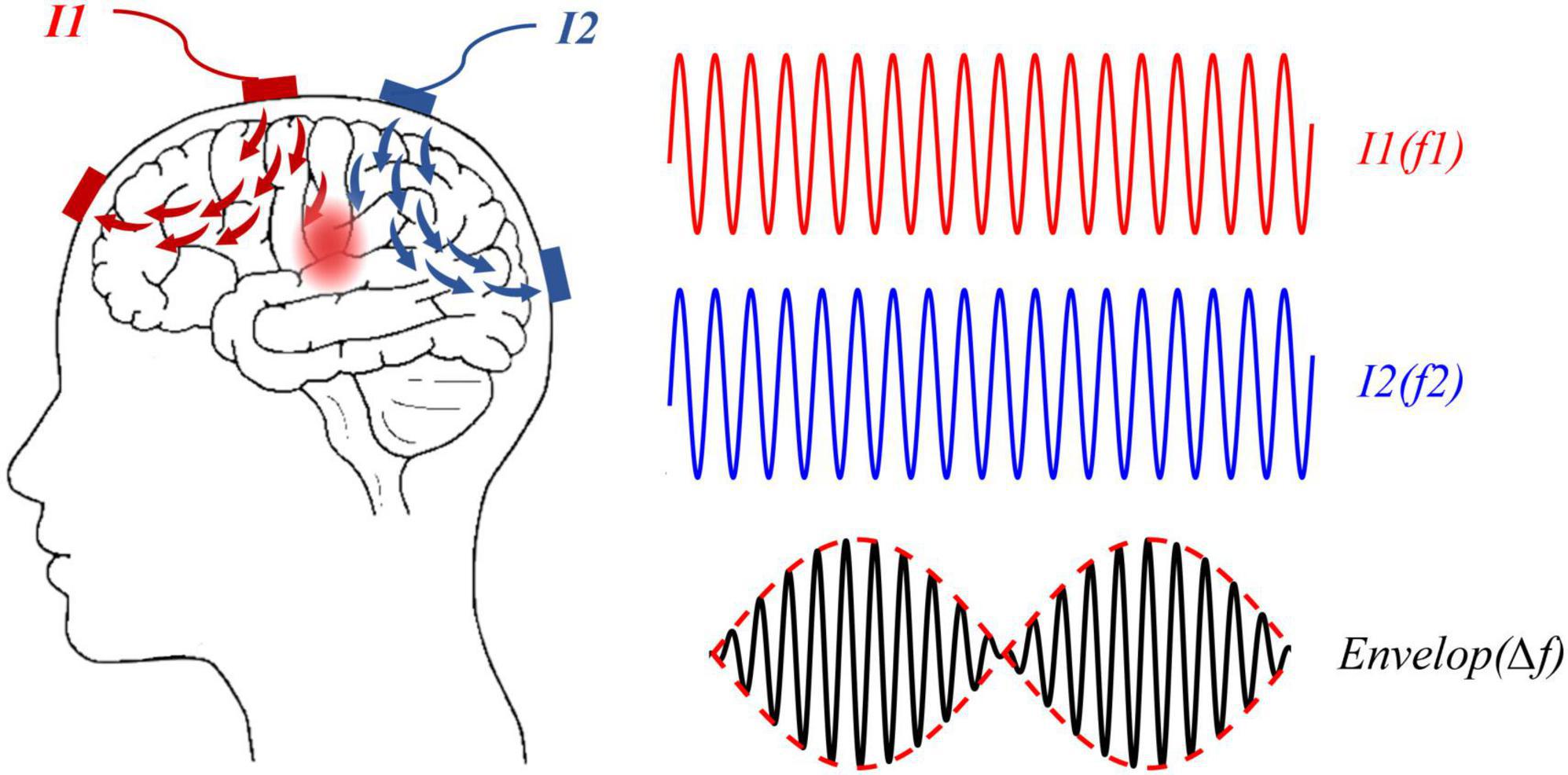
Temporal interference stimulation is based on the delivery of two or more high-frequency electrical currents aimed at deep brain regions. While each individual signal operates at a frequency that is too high to affect neuronal activity directly, the slight difference between their frequencies creates an interference pattern. This results in a low-frequency envelope that is capable of modulating neuronal firing within the target region. In effect, TIS allows the stimulation of deep brain structures with precise control without impacting the superficial cortical layers.
How the Interference Works
The interference phenomenon that underpins TIS can be explained mathematically as follows: when two high-frequency sine waves of frequencies \( f_1 \) and \( f_2 \) are superimposed, they produce an amplitude modulated signal with an envelope frequency of \( |f_1 - f_2| \). This modulation results in a lower frequency component that effectively stimulates neuronal clusters in regions such as the globus pallidus internus (GPi) or the subthalamic nucleus (STN), areas implicated in motor control.
Advantages over Traditional Deep Brain Stimulation (DBS)
One of the major challenges in treating PD is balancing efficacy with risk. Traditional DBS, while effective, involves invasive surgical implantation of electrodes, which presents potential complications related to surgery, anesthesia, and long-term hardware management. TIS, on the other hand, offers several distinct advantages:
- Non-Invasiveness: Unlike DBS, TIS does not require any incision or insertion of electrodes, thereby reducing the risk of infection and other surgical complications.
- Targeted Stimulation: By focusing on deep brain regions without stimulating the overlying tissue, TIS minimizes side effects and allows for more precise modulation of affected neural circuits.
- Early Intervention Potential: The non-invasive nature of TIS could make it a viable option for patients in the early stages of PD, potentially delaying the progression of motor symptoms.
Clinical Evidence and Pilot Studies
Pilot Clinical Studies
Recent pilot studies have provided preliminary evidence that TIS can lead to measurable improvements in patients with mild PD. In one notable study, participants experienced a reduction in overall Parkinson’s Disease severity by an average of 15% after a short session of TIS. Improvements were particularly significant in the reduction of bradykinesia and tremors.
Key Findings from Clinical Trials
The application of TIS in a controlled pilot setting has yielded several important observations:
- Improved Motor Scores: Patients exhibited measurable enhancements on the Movement Disorder Society-revised Unified Parkinson's Disease Rating Scale (MDS-UPDRS-III), a scale used to evaluate the motor symptoms in PD.
- Enhanced Neuronal Regulation: The low-frequency envelope generated by TIS appears to recalibrate disrupted neuronal circuits involved in motor control, resulting in better coordinated movement patterns.
- Patient Tolerability: The non-invasive procedure was well tolerated by patients, with minimal discomfort and a favorable side-effects profile when compared to more invasive techniques.
Evidence Table
The following table summarizes the key elements comparing traditional DBS and TIS in terms of application, efficacy, and safety:
| Parameter | Deep Brain Stimulation (DBS) | Temporal Interference Stimulation (TIS) |
|---|---|---|
| Invasiveness | Surgical implantation; high risk of procedural complications | Non-invasive; reduced risk, no surgery |
| Target Specificity | Precise but affected by electrode placement issues | Focal stimulation using interference patterns targeting deep regions |
| Patient Accessibility | Limited to advanced cases due to surgical risks | Potentially applicable in early stages |
| Side Effects | Risk of surgical and long-term hardware-related complications | Minimal side effects reported; better overall tolerability |
| Adaptability | Limited adaptability post-surgery | High flexibility with dynamic adjustment of stimulation parameters |
Detailed Mechanism of Temporal Interference Stimulation
Electrical Field Dynamics
At the core of TIS is the understanding that neurons can be selectively activated by electrical fields when the stimulus is within a particular frequency range. By utilizing two separate high-frequency currents with slight frequency differences, TIS creates an interference pattern deep within the brain. This pattern forms a beat frequency, which falls within the ideal stimulatory range for neurons involved in motor control. The primary advantage of this approach is that it allows clinicians to modulate the stimulation intensity and frequency in real time, tailoring the therapy to the patient’s immediate neurological responses, thereby optimizing outcomes.
Technical Considerations
The efficacy of TIS relies on precise calibration of the electrode placements and the frequency difference between the applied currents. For instance, if the two currents are set at frequencies \( f_1 \) and \( f_2 \), the neuronal stimulation occurs at the difference \( \Delta f = |f_1 - f_2| \). This low-frequency envelope is critical as it mimics the natural firing rhythms of brain regions responsible for motor control. Careful adjustment of these parameters is essential for maximizing therapeutic benefits while avoiding unintended neural activation in off-target regions.
Comparative Benefits for Parkinson's Disease
The symptomatic improvements observed in PD patients with TIS are primarily due to its ability to restore balanced neural activity in disrupted motor circuits. The technique has been particularly effective in mitigating two of the most challenging symptoms:
- Tremors: Uncontrolled shaking in PD patients can be reduced by stabilizing the neuronal circuits in the GPi and STN, which are often hyperactive in tremor conditions.
- Bradykinesia: The slow movement characteristic of PD is linked to insufficient activation of motor circuits. TIS increases neuronal firing in these pathways, thereby enhancing motor responsiveness.
These improvements emphasize TIS's ability to target deep brain structures that are critical to motor function regulation. With further adaptation, it may offer a broader application in PD symptom management, potentially delaying the need for more invasive procedures.
Challenges and Future Directions
Current Limitations
Although the early results with TIS are promising, there are several challenges that need to be addressed before it can become a mainstream treatment option for Parkinson’s Disease:
- Limited Sample Sizes: Most clinical trials so far have involved small cohorts. Larger, randomized controlled trials are necessary to validate the preliminary observations and ensure consistent efficacy across diverse patient populations.
- Parameter Optimization: There is a need for further research into the optimal frequency, intensity, and duration of the stimulation sessions. Fine-tuning these parameters is crucial for maximizing patient benefit while ensuring safety.
- Long-term Effects: The long-term impact of repeated TIS sessions on brain plasticity and overall neural health is still largely unknown. Extended trials will help clarify the durability and safety of sustained TIS use.
- Individual Variability: Variations in individual anatomy and disease progression mean that a one-size-fits-all approach is unlikely. Personalized treatment plans based on patient-specific neurophysiological characteristics are expected to yield the best outcomes.
Research and Development Pathways
To address these limitations, the future research agenda for TIS includes:
- Expanded Clinical Trials: Initiatives aimed at enrolling larger numbers of participants can provide more robust statistical evidence regarding TIS efficacy and safety in PD treatment.
- Technological Enhancements: Improvements in electrical stimulation equipment, electrode design, and real-time monitoring systems are expected to enhance the precision of TIS delivery.
- Multidisciplinary Collaborations: Integrating expertise from neurology, biomedical engineering, and data science can lead to innovative approaches in parameter optimization and therapy customization.
- Exploring Combined Therapies: Investigations into combining TIS with other advanced neuromodulatory techniques—such as nanoparticle-assisted drug delivery or focused ultrasound—could open new avenues for synergistic treatment strategies aimed at mitigating PD symptoms.
Additionally, research into adaptive algorithms that continuously monitor a patient’s neural response and adjust the stimulation parameters might offer a pathway toward real-time personalized therapy. Such an approach would be a significant leap forward in the non-invasive treatment of PD, ensuring that therapeutic interventions remain finely tuned to the evolving disease state of each patient.
Comparative Analysis with Existing Treatments
Traditional vs. Non-Invasive Approaches
The landscape of Parkinson’s disease treatment has been dominated by pharmacological therapies and invasive procedures like DBS. While medications such as levodopa can temporarily relieve motor symptoms, they often lead to complications like dyskinesia with prolonged use. DBS, while effective in managing advanced symptoms, carries substantial procedural risks and a high cost of care. Temporal interference stimulation stands out by offering a less invasive alternative that does not compromise on the targeted stimulation of deep brain structures.
A comparative review of these techniques reveals several critical factors:
- Surgical Risk: The invasive nature of DBS requires strict patient selection, limiting its use to individuals with advanced PD. In contrast, TIS minimizes the risk associated with invasive surgery, making it a potential option for earlier intervention.
- Adaptability of Treatment: TIS provides the possibility for dynamic adjustment of stimulation protocols, an advantage over the relatively static settings used in traditional DBS once electrodes are implanted.
- Accessibility: Reduced invasiveness not only lowers the risk profile but may also result in lower overall healthcare costs and greater patient accessibility, particularly in settings where surgical options are limited.
In summary, TIS represents a significant advancement in neurotechnology by potentially combining the efficacy of deep brain stimulation with the safety and adaptability of non-invasive treatment approaches. Its ability to target specific neural circuits with minimal collateral effects makes it a subject of high interest for further clinical exploration.
Conclusion and Final Thoughts
Temporal interference stimulation emerges as a compelling non-invasive approach to managing motor symptoms associated with Parkinson's Disease. The underlying mechanism, which utilizes high-frequency electrical currents to create a beat frequency that precisely targets deep brain structures, offers a fresh perspective on neuromodulation. Clinical pilot studies have shown promising results, with significant reductions in tremor and bradykinesia alongside improvements in overall motor function scores. Importantly, the non-invasive nature of TIS opens the possibility for early intervention in PD, potentially slowing disease progression without the risks inherent in surgical procedures such as DBS.
Several advantages—including the ability to modulate stimulation parameters in real-time, the targeted reduction of motor symptoms, and high patient tolerability—position TIS as a revolutionary treatment avenue pending further validation through larger-scale clinical trials. Challenges remain, especially regarding optimal parameter settings, long-term effects, and individual variability in treatment response. Therefore, ongoing research is essential to establish standardized protocols and assess the durability of clinical benefits.
Overall, TIS reinforces the evolving paradigm of precision neuromodulation. It not only enhances our capability to intervene non-invasively in the neurological pathways underlying Parkinson's Disease but also embodies the shift toward more adaptive and personalized therapeutic strategies in the broader field of neurology.
References
- Article on Temporal Interference Stimulation - Nature
-
Study on TIS in Parkinson's Disease - PubMed
- TIS Pilot Study in PD - BrainStimulation Journal
- Frontiers in Human Neuroscience Article on TIS
- Using Beat Patterns to Treat Parkinson's Disease
Recommended Further Queries
Last updated February 27, 2025